|
4/3/2024 0 Comments Thomson atomic theory 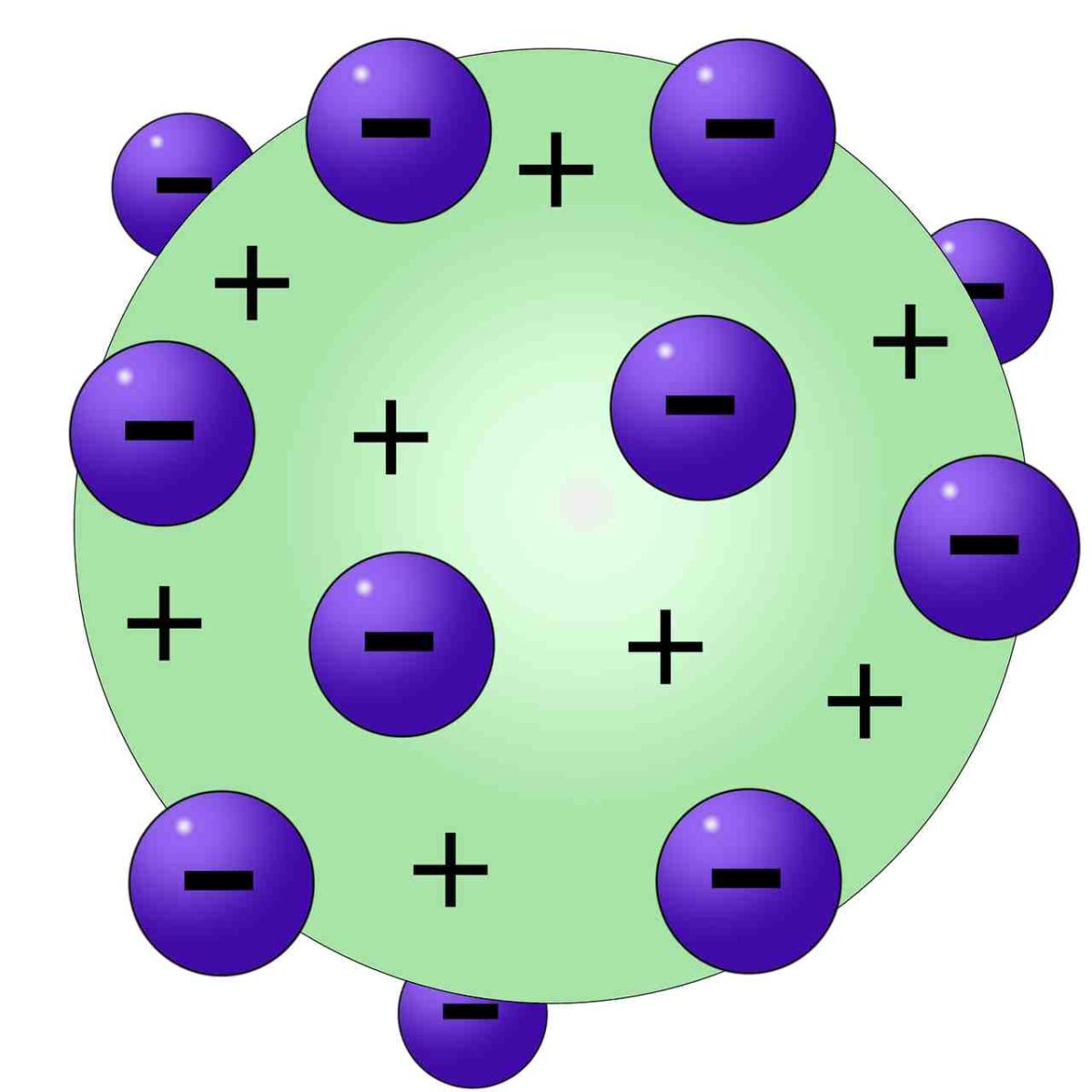
He could then calculate the charge-to-mass ratio (e/m) of the electron. The magnetic field deflected the electrons into circular paths of known radius (r). Since the charge of an electron was now known due to Millikan’s research, and the charge-to-mass ratio was already known due to Thomson’s research (1. Thomson used an electric field (V) to accelerate electrons into a magnetic field (B). Millikan concluded that this value must therefore be a fundamental charge-the charge of a single electron-with his measured charges due to an excess of one electron (1 times 1.6 10 −19 C), two electrons (2 times 1.6 10 −19 C), three electrons (3 times 1.6 10 −19 C), and so on, on a given oil droplet. Atoms of different elements differ in size, mass, and other properties. Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed. The general tenets of this theory are: All matter is composed of extremely small particles called atoms. Thomson proposed a model of the atom in 1904 after doing a series of cathode tube experiments. Dalton proposed his atomic theory in 1804. The first seed of the model we are discussing appear in his famous 1897 announcement of the discovery of the electron. Following his discovery of the electron in 1897, British Physicist J.J. Thomson had been in the business of proposing atomic models since at least 1881, which is when he proposed his 'vortex' model of the atom. The idea that electricity is transmitted by a tiny particle related to the atom was first forwarded in the 1830s. He also researched colour blindness, which he had as a result, colour blindness is known as Daltonism in several languages. 1 He introduced the atomic theory into chemistry. Looking at the charge data that Millikan gathered, you may have recognized that the charge of an oil droplet is always a multiple of a specific charge, 1.6 10 −19 C. Leadup to Thomsons 1904 Model of the Atom. John Dalton FRS ( / dltn / 5 or 6 September 1766 27 July 1844) was an English chemist, physicist and meteorologist. The tabulated data are examples of a few possible values. \): Millikan’s experiment measured the charge of individual oil drops. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
